![]() Figure 9 of
Perkins, Mol Vis 2003;
9:60-73.
Figure 9 of
Perkins, Mol Vis 2003;
9:60-73.
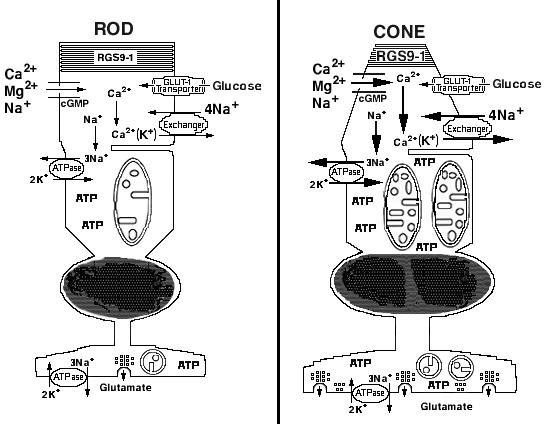
Figure 9. Light-adapted rod and cone photoreceptor
Schematic drawing of light-adapted rod and middle wavelength-sensitive (M) cone photoreceptors illustrating fundamental bioenergetic and functional differences. Outer segment: The relatively larger arrows in the cone, compared to rod, outer segments indicate the cone's higher relative permeability for Ca2+ through the cGMP-gated channel, higher fraction of the dark current carried by Ca2+ and more rapid Na+/Ca2+(K+) exchanger [15-18]. The GLUT-1-type glucose transporter appears to have similar kinetics in rod and cone outer segments [5,72]. The relatively larger lettering for RGS9-1 in cone outer segments indicates the higher content of this GTPase accelerating protein (GAP) in mouse cones, compared to rods [21,64]. Inner segment: The relatively larger arrows in the cone, compared to rod, inner segment Na+,K+-ATPase indicates the higher relative activity of the cones [20]. We found that mouse rods and cones in situ are in the orthodox, rather than the condensed, conformation (see text and Hackenbrock [30]). The mouse rods have an average of approximately 5 mitochondria per cell, whereas cones have approximately 10 mitochondria per cell (see text and Figure 1B). Mouse cone mitochondria also were more uniformly stained and more reactive for cytochrome c oxidase than rod mitochondria (see text and Figure 1B). Consistent with our results, cones also contain more ATP than rods [59]. The cone mitochondria had narrower crista junctions, greater cristae connectivity and approximately 3-fold more cristae membrane surface area compared to rods (see text and Figure 4, Figure 5, and Figure 6). Synaptic terminal: The rod synaptic terminal or spherule contains a single invagination of the plasma membrane where glutamate is released. The spherule contains one or two small mitochondria. The cone synaptic terminal or pedicle contains numerous invaginations as well as flat contact sites where glutamate is released. The pedicle contains several medium to large mitochondria [1,4,6-8]. The cone mitochondria are more reactive for cytochrome c oxidase than the rod mitochondria ([9,20]; unpublished data).