![]() Figure 3 of
Morin, Mol Vis 2003;
9:449-459.
Figure 3 of
Morin, Mol Vis 2003;
9:449-459.
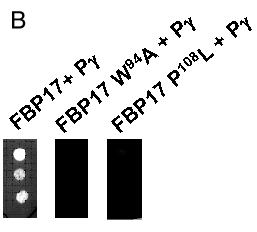
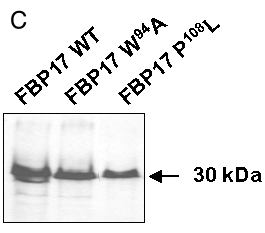
Figure 3. Mutations in FBP17 SH3 domain interrupt binding to Pγ-rod
A: Sequence of the FBP17 clone with the SH3 domain in lowercase letters: tryptophane-94 and proline-108 residues, mutated respectively into alanine (W94A mutant) and leucine (P108L mutant), are in red. B: The indicated mutants of FBP17 (as gal4AD fusions) were transformed into L40 yeast expressing the lexADBD-Pγ-rod bait and plated as triplicate drops on medium lacking histidine. Positive control corresponds to wild-type FBP17. Results were confirmed on three independent clones for each mutation. C: Protein extracts from the cotransformants described above were analyzed by western blot to verify that wild type and mutated forms of FBP17 were correctly expressed. The gal4AD-FBP17 fusion proteins were detected with anti-hemagglutinin antibody as described in "Experimental procedures".
A:
FBP17: RQSGLYDGQT HQTVTNCAQD RESPDGSYTE EQSQESEHKV LATDFDDEFD DEEPLpaigt ckalytfegq FBP17: negtisvveg etlsvieedk gdgwtrirrn edeegyvpts yvevyldKNA KGS 123 |