![]() Figure 5 of
Saleh, Mol Vis 2001;
7:240-246.
Figure 5 of
Saleh, Mol Vis 2001;
7:240-246.
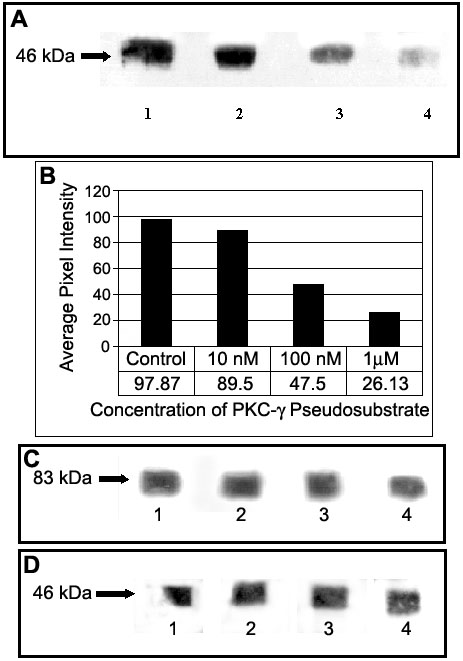
Figure 5. Inhibition of serine phosphorylation on Cx46
Inhibition of serine phosphorylation on Cx46 by myristoylated pseudosubstrate inhibitory peptide against PKC-g in the lens cortex. Fiber cells of the lens cortex (3 lenses/sample) were dissected and incubated with different concentrations of myristoylated pseudosubstrate peptide specific for PKC-g. Then, Cx46 was immunoprecipitated and the samples were analyzed by SDS-PAGE and western blot. The transferred proteins on western blots were probed with anti-phosphoserine antibodies. A: Western blots showing phosphorylated serine on Cx46 after treatment with different concentrations of PKC-g pseudosubstrate inhibitory peptide (lane 1: Control, no pseudosubstrate peptide, designated as the 100% reference; lane 2: 10 nM peptide; lane 3: 100 nM peptide; lane 4: 1 mM peptide). B: Quantitative analysis of inhibition of serine phosphorylation on Cx46 in the lens cortex by PKC-g pseudosubstrate inhibitory peptide. C: Western blots from A containing immunoprecipitated Cx46 were probed with anti-PKC-g to determine that PKC-g pseudosubstrate did not inhibit the interaction between Cx46 and PKC-g. D: As a control, Western blots from A were probed with anti-Cx46 to demonstrate equal loading of immunoprecipitated Cx46.