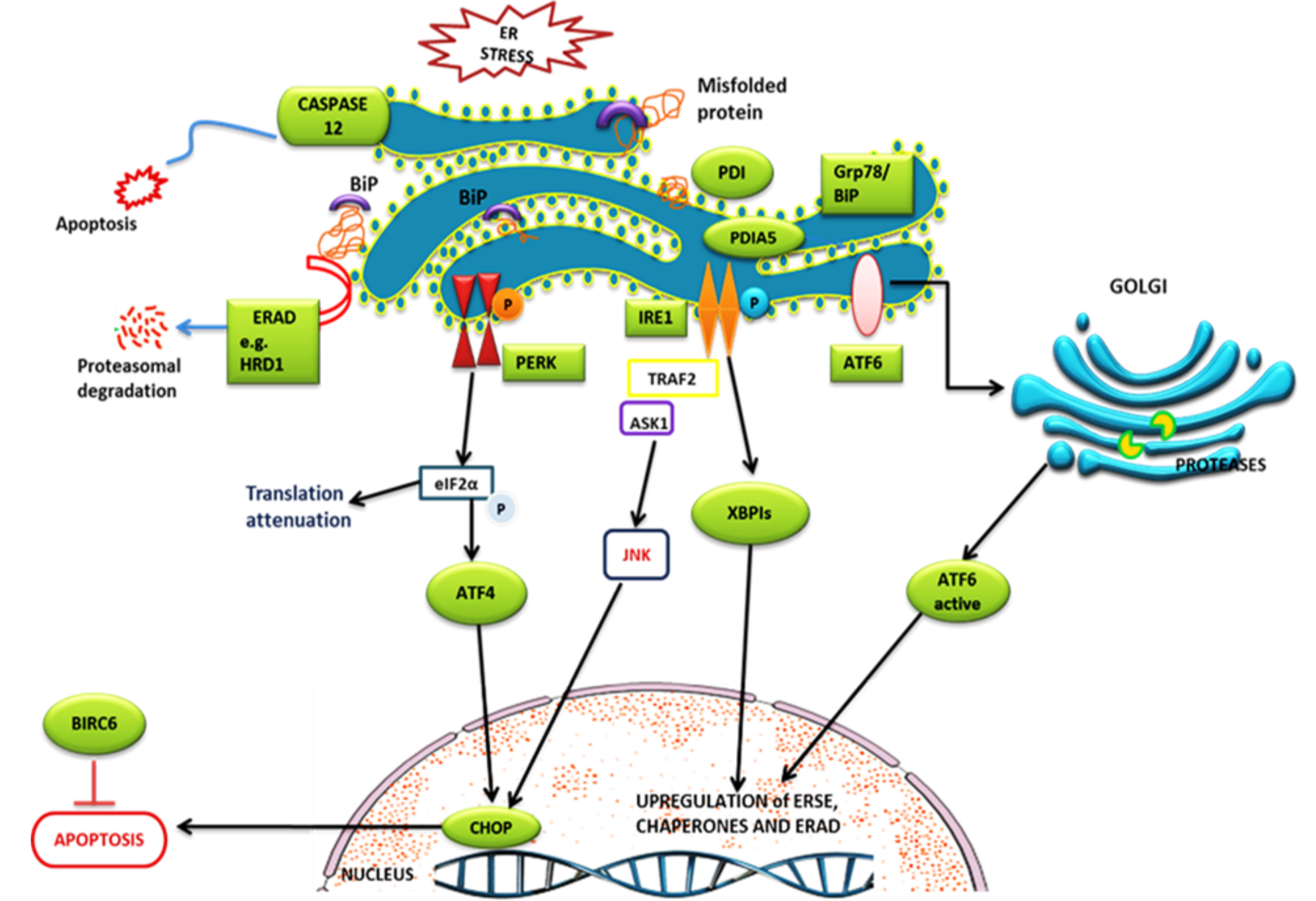
Figure 4. Schematic representation of the UPR signaling pathway. The unfolded protein response (UPR) is activated by the accumulation
of misfolded proteins in the endoplasmic reticulum (ER) and is mediated by three key branches: inositol-requiring enzyme 1
(IRE1), activated transcription factor 6 (ATF6), and Protein kinase RNA-like ER kinase (PERK), all regulated by glucose-regulated
proteins 78/Binding protein (GRP78/BiP). Upon ER stress, GRP78 dissociates from these sensors, leading to their activation.
PERK phosphorylates elongation factor 2α (eIF2α), temporarily halting global protein synthesis to reduce ER burden. IRE1 splices
X-box binding protein 1 (XBP1) mRNA, producing a transcription factor that enhances protein folding and processing, while
ATF6 translocate to the Golgi for cleavage into its active form, upregulating ER stress genes like GRP78. Anti-apoptotic factors,
baculoviral inhibitor of apoptosis protein containing 6 (BIRC6) and HRD1 (Synoviolin) facilitate the degradation of pro-apoptotic
proteins, promoting cell survival. However, prolonged ER stress leads to activation of JNK via IRE1 and PERK, triggering caspase-dependent
apoptosis, while CCAAT-enhancer-binding homologous protein (CHOP) and CASPASE12 further contribute to cell death [
81-
85]. The cell organelles template was adapted from Biorender.com, Toronto, Canada.
 Figure 4 of
Hayat, Mol Vis 2025; 31:463-484.
Figure 4 of
Hayat, Mol Vis 2025; 31:463-484.  Figure 4 of
Hayat, Mol Vis 2025; 31:463-484.
Figure 4 of
Hayat, Mol Vis 2025; 31:463-484.