Figure 6. Structure-based analysis of the effect of the L750-K751 deletion. CPP12, linker, and CPP13 colored in green, purple and cyan,
respectively. The structure is shown in cartoon, with the linker side chain shown in sticks; residues in the two CPP domains
that participate in the formation of the hydrophobic patch are shown in spheres. A: Structural model of wt after loop remodeling, highlighting the central position of L750. B: Corresponding model of the deletion mutant: in the shortened loop, D748 replaces L750, perturbing the hydrophobic character
of this region and leading to destabilization. C, D: Different relative orientations adapted by the CPP domains in wt (C) and deletion mutant (D) indicate increased variability in orientation for the deletion mutant compared to the wt.
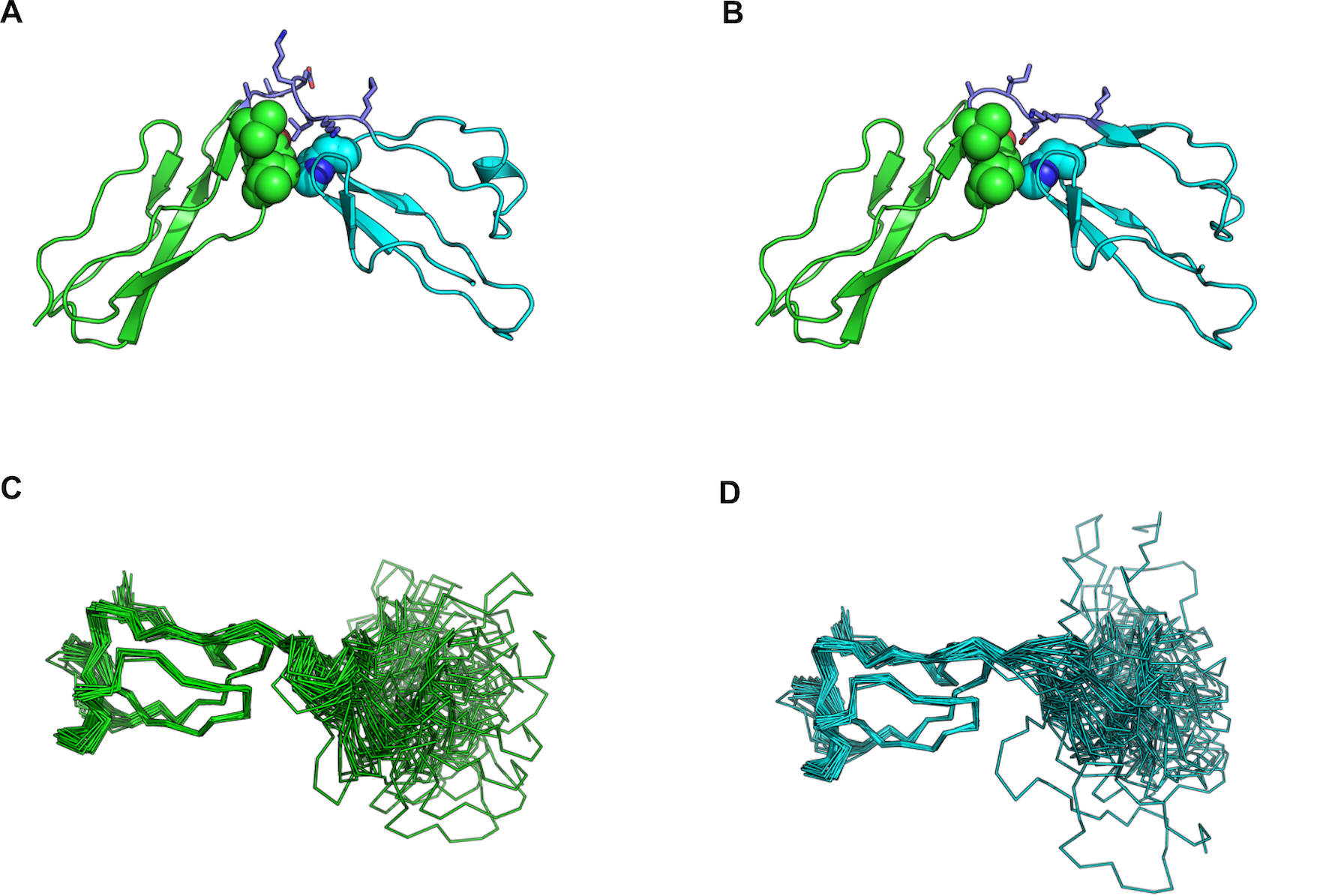
 Figure 6 of
Namburi, Mol Vis 2020; 26:299-310.
Figure 6 of
Namburi, Mol Vis 2020; 26:299-310.  Figure 6 of
Namburi, Mol Vis 2020; 26:299-310.
Figure 6 of
Namburi, Mol Vis 2020; 26:299-310. 