Figure 1. Confirmation of recombinant αA-crystallin wild-type and mutants. A: The 2% agarose gel electrophoresis image shows confirmation of clones by DNA double-digestion using the restriction enzymes
NcoI and BamHI. The double-digested products show two individual bands approximately 4.6 kb and 512 bp representing the vector
and the insert, respectively. Lanes 2–5 denote the doubly digested product of R12C, R21L, R49C, and R54C, respectively. M1
is the 100 bp DNA ladder and M2 1 kb DNA ladder. B: The boxed chromatogram represents the site-directed mutation at the specific points (circled) in the αA-crystallin gene.
The circle represents the change in the nucleotide that creates N-terminal crystallin mutants such as R12C, R21L, R49C, and
R54C. The second chromatogram represents αA-crystallin wild-type (wt). C–D: Sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS–PAGE) and western blotting, respectively, of the αA-crystallin
wt and N-terminal mutants. The purified αA-crystallin wt and N-terminal mutants (R12C, R21L, R49C, and R54C) were separated
by 15% SDS–PAGE and transferred on to the nitrocellulose membrane for blotting. The blot was probed with human anti-αA-crystallin
antibody, followed by secondary anti-rabbit immunoglobulin G (IgG) antibody conjugated to alkaline phosphatase. The blot was
developed by adding the 5-Bromo-4-Chloro-3-idolyl Phosphate / Nitroblue tetrazolium chloride (BCIP/NBT) substrate. The appearance
of insoluble dark blue diformazan precipitate in the blot confirmed the presence of αA-crystallin.
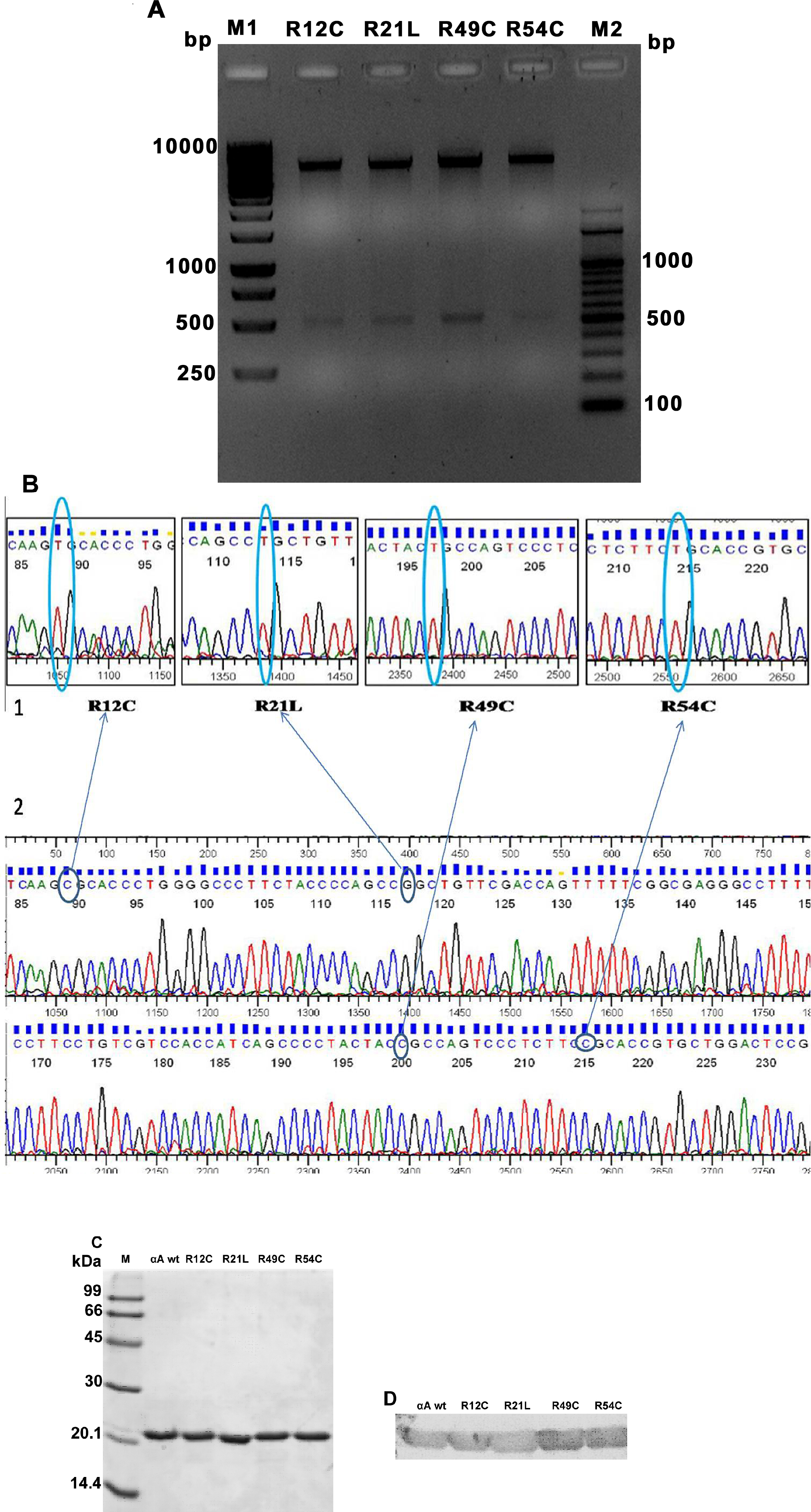
 Figure 1 of
Ramkumar, Mol Vis 2014; 20:1002-1016.
Figure 1 of
Ramkumar, Mol Vis 2014; 20:1002-1016.  Figure 1 of
Ramkumar, Mol Vis 2014; 20:1002-1016.
Figure 1 of
Ramkumar, Mol Vis 2014; 20:1002-1016. 