Figure 5. Analysis of the xlProminin-1
alternatively spliced isoforms in four tissues: the retina, brain,
testis, and kidney. We found that profiles of the xlProminin-1
alternative splicing are different in these tissues. A:
Products of reverse-transcription PCR (RT–PCR) from a region of
extensive alternative splicing on the xlProminin-1 gene (exon 20 to 28)
were separated on a 1% agarose gel and visualized with ethidium bromide
staining. Five discrete PCR products were excised from the gel, cloned,
and sequenced. Their exon compositions were determined and each product
was designated with a Greek letter (α, β, γ, δ, and ε) for
identification when used in the quantitative analysis shown in panel B.
The predominant isoforms of xlProminin-1 expressed in the retina lack
the alternatively spliced exons 26b, 27, and 28a, whereas the
predominant isoform of xlProminin-1 expressed in the kidney retains
these exons. The predominant isoforms of xlProminin-1 expressed in the
brain and kidney retain exon 28a. Exon 27 was retained only when exon
26b was retained as well. B: Quantification of the xlProminin-1
alternatively spliced isoforms in four tissues: the retina, brain,
testis, and kidney. The resolved RT–PCR products were linearly scanned
and the optical densities were plotted. Green arrows indicate peaks
that represent isoforms resulting from alternative splicing. The major
isoform of xlProminin-1 expressed in the retina, ε, lacks the
alternatively spliced exons 26b, 27, and 28a, whereas the major form of
xlProminin-1 expressed in the kidney, α, retains all possible exons.
The majority of xlProminin-1 expressed in the brain and kidney retains
exon 28a.
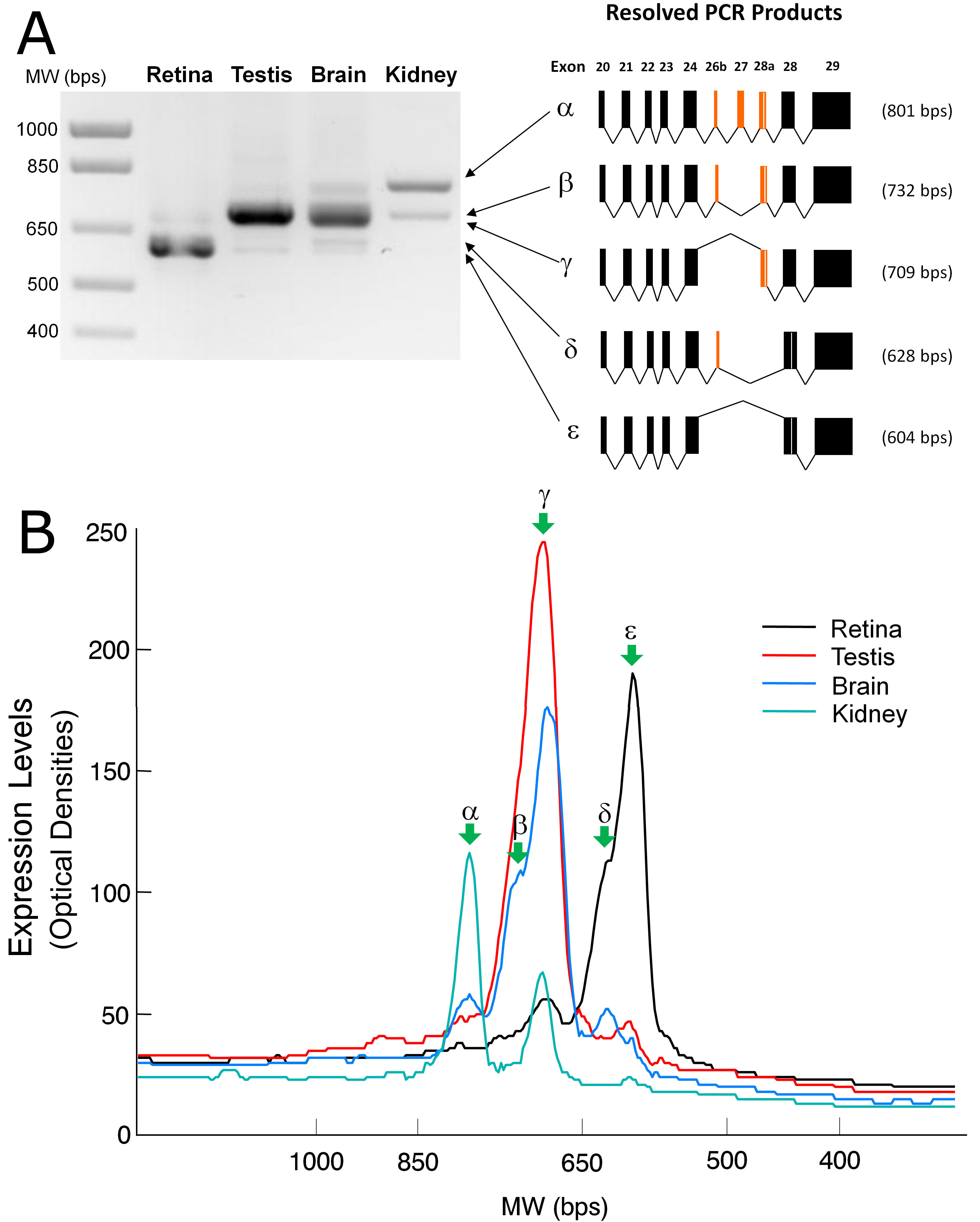
 Figure 5 of Han, Mol Vis 2011; 17:1381-1396.
Figure 5 of Han, Mol Vis 2011; 17:1381-1396.  Figure 5 of Han, Mol Vis 2011; 17:1381-1396.
Figure 5 of Han, Mol Vis 2011; 17:1381-1396. 