Figure 8. Chaperone-like activity of
αA-crystallin and G98R αA-crystallin with and without Cu2+
using insulin and citrate synthase as target proteins. The difference
in the chaperone-like activity of the mutant protein with respect to
the wild type protein toward DTT-induced aggregation of insulin at
37 °C and heat-induced aggregation of CS at 43 °C was assayed
in the absence and the presence of Cu2+. A:
Aggregation of 0.2 mg/ml insulin (In) in 10 mM phosphate buffer (pH
7.4) containing 100 mM NaCl was monitored by light scattering at 465 nm
(expressed in arbitrary units [AU]) in the absence or in the presence
of 0.1 mg/ml αA-crystallin and G98R αA-crystallin. A similar experiment
was performed in the presence of 15 μM Cu2+. B:
Aggregation of 25 μg/ml citrate synthase (CS) was monitored by light
scattering at 465 nm in the absence and in the presence of
20 μg/ml of either αA-crystallin or G98R αA-crystallin. C:
The effect of αA-crystallin, G98R αA-crystallin, and their mixed
oligomer on the aggregation of CS in the presence of 3 μM Cu2+ was
measured. D: Percentage protection of CS aggregation in the
presence of 1 μM αA-crystallin (○) and G98RαA-crystallin (●) as a
function of Cu2+ concentration indicate that the intrinsic
chaperone ability of αA-crystallin is increased and that of G98R
αA-crystallin is decreased. The experiments were performed three times,
and the trends were reproducible. Representative data are shown.
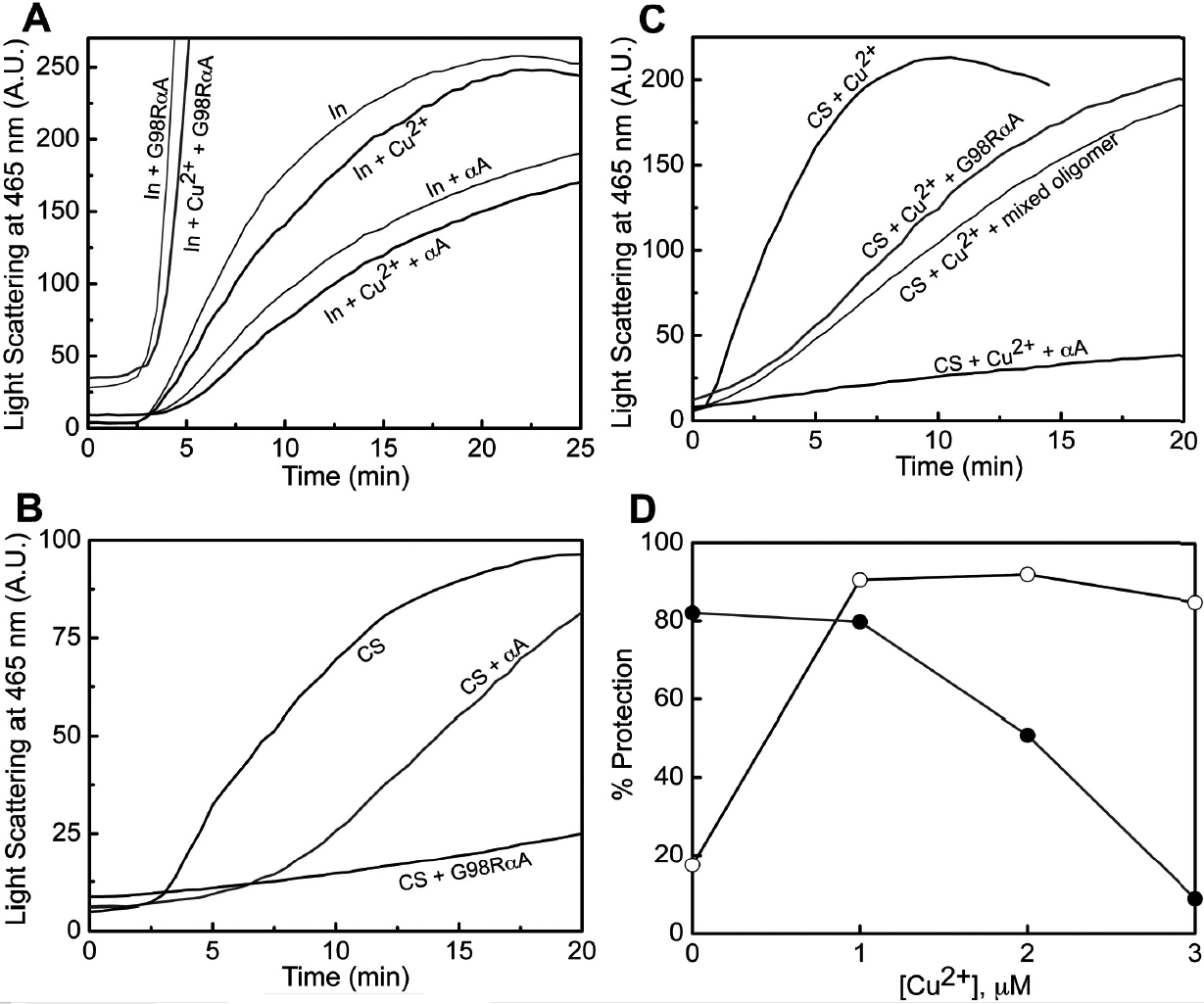
 Figure 8 of Singh, Mol Vis 2009; 15:2050-2060.
Figure 8 of Singh, Mol Vis 2009; 15:2050-2060.  Figure 8 of Singh, Mol Vis 2009; 15:2050-2060.
Figure 8 of Singh, Mol Vis 2009; 15:2050-2060. 