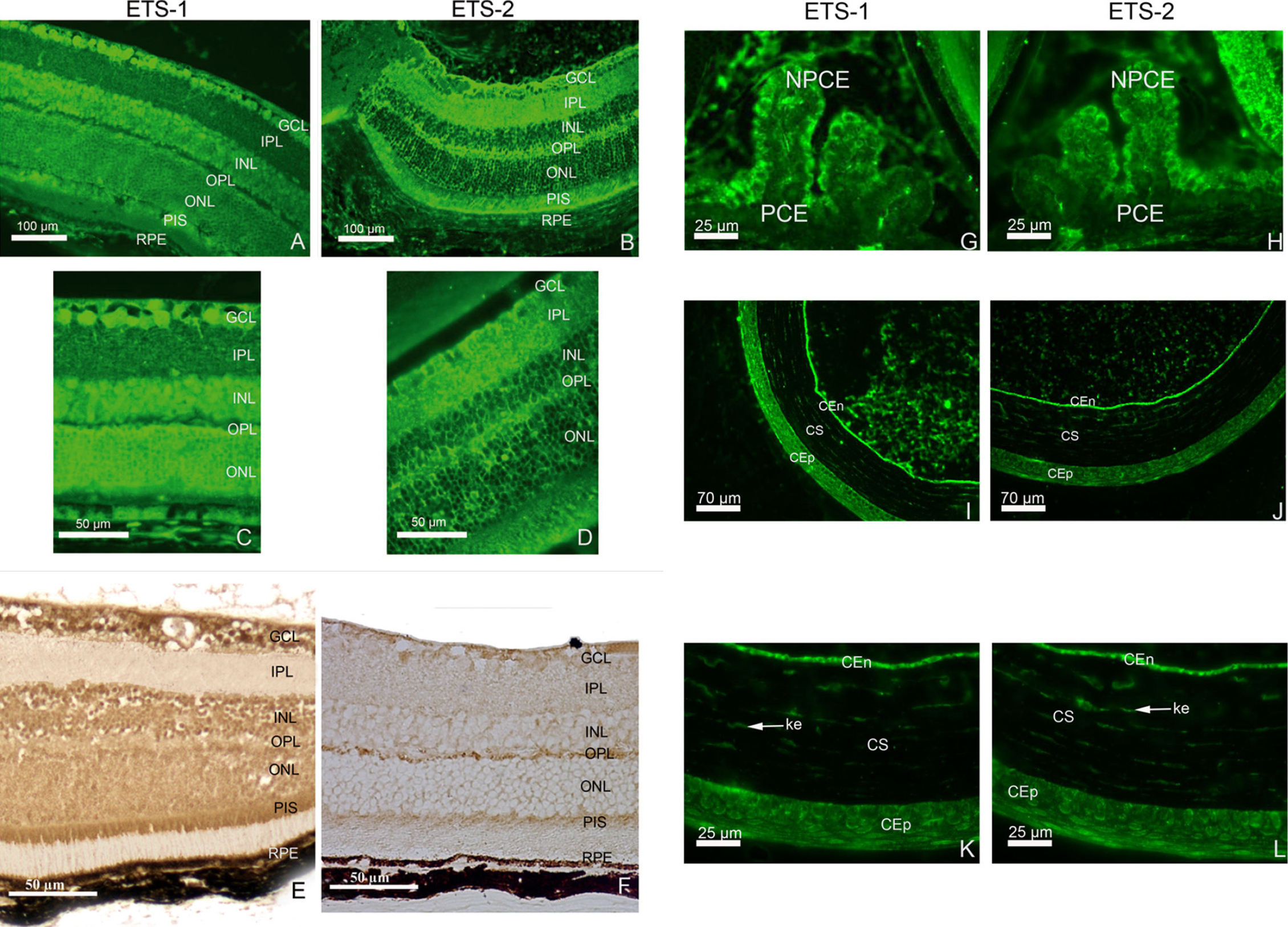
Figure 2. Distribution of ETS
transcription factor proteins in mouse adult retina, cornea, and
ciliary body as determined by immunohistofluorescence.
Immunohistofluorescence for ETS-1 and ETS-2 is shown (A-D).
ETS-1 (A and C) is present in the ganglion cell layer
(GCL) and inner nuclear layer (INL) with intense homogeneous staining
in the outer nuclear layer (ONL). It is also detected in the
photoreceptor inner segment (PIS) and retinal pigment epithelium (RPE).
ETS-2 (B and D) is also present in significant amounts
in the GCL. Weak ETS-2 immunoreactivity is observed in the INL and ONL.
In these layers, at higher magnification, very few cell bodies are
immunostained. The immunolabeling detected seems to be restricted to
the nuclear membranes whereas the nucleoplasm seems to be devoid of
ETS-2 immunolabeling. In contrast to what was observed for the retinal
distribution of ETS-1 immunoreactivity, strong ETS-2 immunolabeling is
observed in the inner plexiform layer (IPL) and outer plexiform layer
(OPL). As observed for ETS-1, the PIS and RPE display marked ETS-2
immunostaining, although the RPE seems to be less strongly stained for
ETS-2 than for ETS-1. ETS-1 (E) and ETS-2 (F) protein
distributions are shown in the adult retina with the DAB immunostaining
protocol. We observed ETS-1 (G) and ETS-2 (H)
immunolabeling in the adult ciliary body. In the ciliary body, ETS-1
and ETS-2 proteins are detected in the nonpigmented ciliary epithelial
cells (NPCE). No significant labeling is observed in the pigmented
ciliary epithelial cells (PCE). ETS-1 immunolocalisation was detected
in the adult cornea at low (I) and high (K)
magnification. ETS-2 immunolocalisation was detected in the adult
cornea at low (J) and high (L) magnification. In the
cornea, ETS-1 and ETS-2 immunoreactivities are observed in the
epithelium (CEp), the stroma (CS), the stromal keratocytes (ke,
arrows), and the endothelium (CEn).
![]() Figure 2 of De la
Houssaye, Mol Vis 2008; 14:1912-1928.
Figure 2 of De la
Houssaye, Mol Vis 2008; 14:1912-1928. 