Figure 1. Therapeutic effect of
CBA-PEDF-AAV2 on choroidal neovascularization development and antibody
responses to AAV2 capsid following single and sequential intravitreal
injections. A-D: Representative CNV images from mouse
eyes that were untreated (A), received a single intravitreal
(IV) treatment (B), received first IV treatment (C), and
received a second IV treatment (D) of AAV2-PEDF. E: The
therapeutic effect of CBA-PEDF-AAV2 on CNV development was evaluated in
animals that received single IV ocular injections in naïve and
preimmunized mice (5 animals each type of injection), and two
sequential IV injections (15 animals were used). The CNV area was
averaged from 25 laser lesions (from 5 mice) for mice that received a
single IV injection and pre-intramuscular (pre-IM) administration of
the vector, and 75 laser lesions (from 15 mice) for mice that received
two sequential intravitreal injections (2X-IV). F: Antibody
response to AAV2 capsid in animals received a single intramuscular
injection, intravitreal, and two sequential intravitreal injections
overtime. Five animals for each type vector injection were used. Arrow
indicates the second injection time.
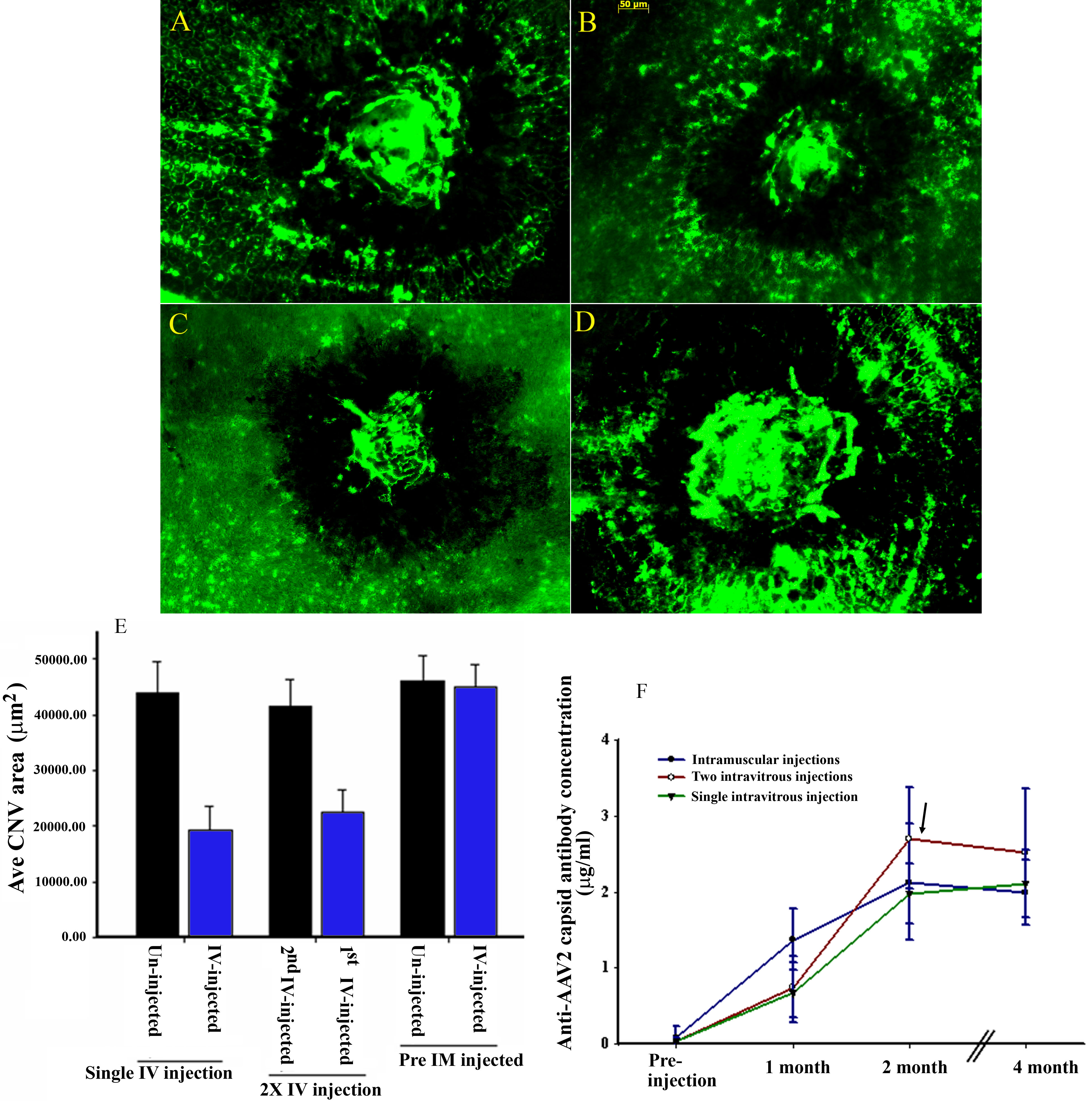
 Figure 1 of Li, Mol Vis 2008; 14:1760-1769.
Figure 1 of Li, Mol Vis 2008; 14:1760-1769.  Figure 1 of Li, Mol Vis 2008; 14:1760-1769.
Figure 1 of Li, Mol Vis 2008; 14:1760-1769. 