![]() Figure 4 of
Fernandes, Mol Vis 2006;
12:1526-1535.
Figure 4 of
Fernandes, Mol Vis 2006;
12:1526-1535.
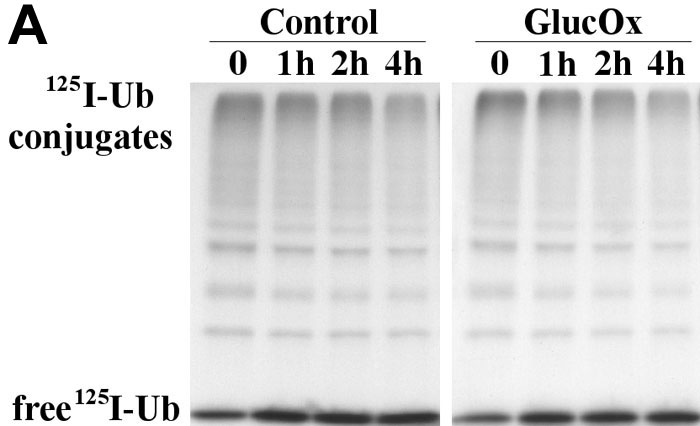
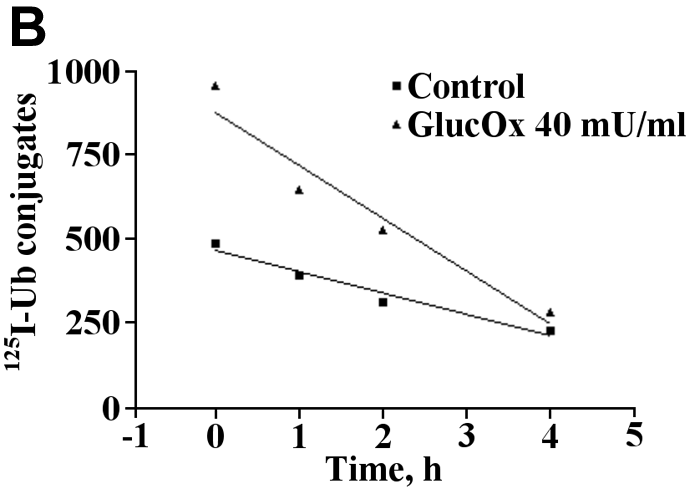
Figure 4. Exposure of endothelial cells to oxidative stress leads to an increased turnover of ubiquitin conjugates
The radiolabeled ubiquitin conjugates were determined by pulse-chase experiments. Lysates from cells previously exposed to glucose oxidase (GlucOx; 40 mU/ml) were pulsed with radiolabeled Ub (125I-Ub) for 30 min (37 °C). Assays were performed in the presence of an ATP-generating buffer and in the presence of an inhibitor of isopeptidases (Ubal). Ubiquitin conjugates were then chased by the addition of an excess of unlabeled Ub. The degradation of 125I-labeled Ub-protein conjugates over time was assessed by SDS-PAGE and autoradiography. The participation of the proteasome in degradation of ubiquitin conjugates was assessed by repeating the same experiment in the presence of 80 μM of proteasome inhibitor MG-132. The difference in the rate of degradation in the presence and absence of MG-132 indicates the contribution of the proteasome for ubiquitin conjugates turnover.