![]() Figure 3 of
Fernandes, Mol Vis 2006;
12:1526-1535.
Figure 3 of
Fernandes, Mol Vis 2006;
12:1526-1535.
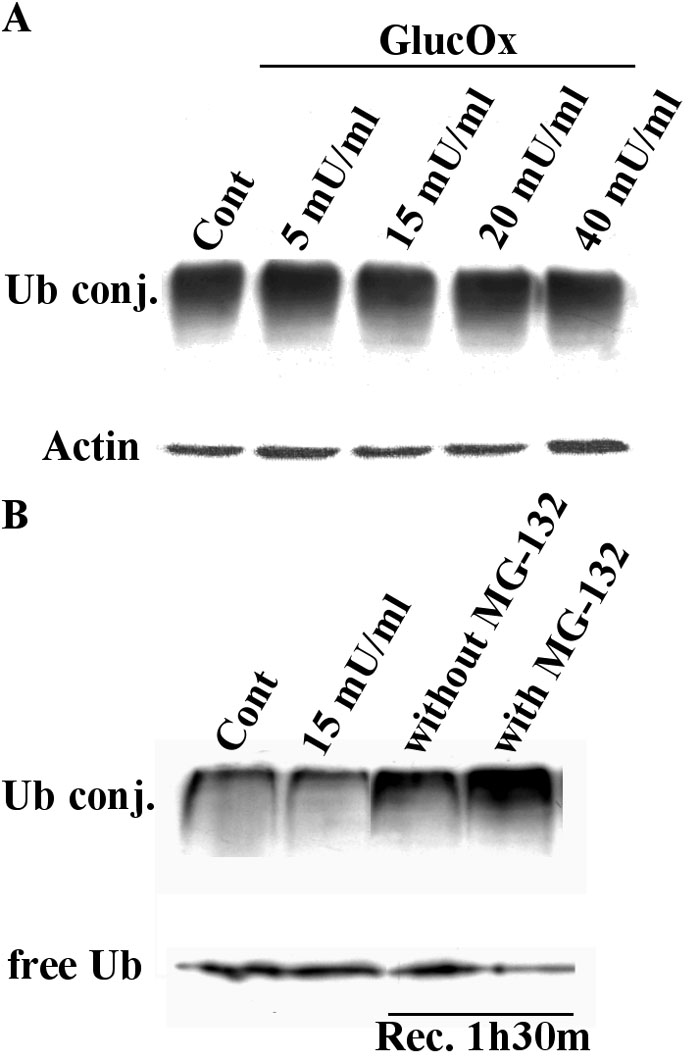
Figure 3. Endogenous ubiquitin conjugates in TR-iBRB cells subjected to oxidative stress
The endogenous ubiquitin conjugates were detected by western blotting. Equal amounts of protein (40 μg) were separated by SDS-PAGE and transferred to polyvinylidene fluoride (PVDF) membranes. A: The ubiquitin conjugates (UbConj) were detected using a monoclonal antibody that recognizes mono- and polyubiquitinylated protein conjugates (FK2). The bottom panel corresponds to actin immunodetection, which was used as an internal loading control. Lane 1, control TR-iBRB cells; Lanes 2-5, TR-iBRB cells treated with increasing concentrations of glucose oxidase (GlucOx), for 4 h. B: Oxidative stress was induced with glucose oxidase for 4 h at 37 °C. Glucose oxidase was then removed, and the cells were allowed to recover from the oxidative stress in the presence or absence of the proteasome inhibitor, MG-132, for 90 min at 37 °C. The detection of free ubiquitin and of ubiquitin protein conjugates was done using a monoclonal antibody directed against ubiquitin. Lane 1, control TR-iBRB cells; Lane 2, TR-iBRB cells treated with 15 mU/ml glucose oxidase for 4 h; Lane 3, recovery from the oxidative stress in the absence of MG-132; Lane 4, recovery from the oxidative stress in the presence of MG-132.