![]() Figure 2 of
Ascano, Mol Vis 2006;
12:1516-1525.
Figure 2 of
Ascano, Mol Vis 2006;
12:1516-1525.
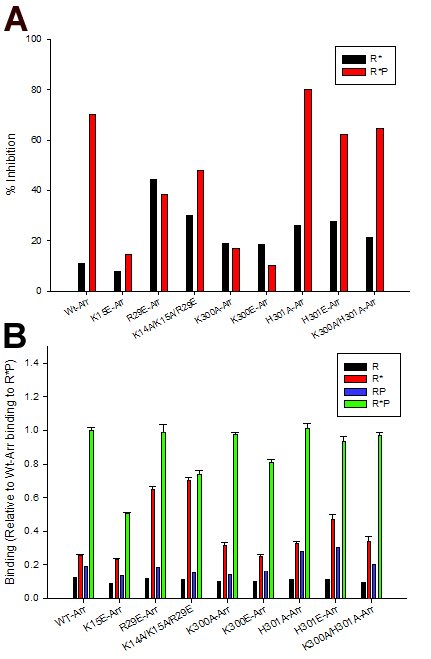
Figure 2. Effect of arrestin mutants on phosphorylated and unphosphorylated rhodopsin
A: Arrestin-mediated rhodopsin deactivation. 35S-γ-GTP assays were performed as described in Methods. WT rhodopsin was incubated either with (gray bars) or without (black bars) rhodopsin kinase. Arrestin was then added to a final concentration of 5 μM. Bars are averages from two independent experiments. B: Direct binding of arrestin mutants to different phosphorylation and photoactivation states of rhodopsin. The ability of the different arrestin mutants to bind to rhodopsin was determined using co-precipitation studies. Arrestin mutants (4 μM) were incubated with membranes from bovine rod outer segments that contained unphotolyzed unphosphorylated (R), photolyzed unphosphorylated (R*), unphotolyzed phosphorylated (RP) and photolyzed phosphorylated (R*P) rhodopsin (7 μM). Error bars represent SEM (n=4). All binding was normalized to binding of WT-Arr to R*P.