![]() Figure 3 of
Rao, Mol Vis 2004;
10:112-121.
Figure 3 of
Rao, Mol Vis 2004;
10:112-121.
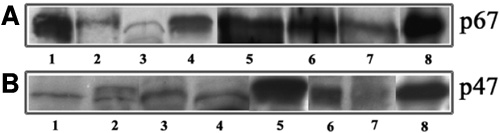
Figure 3. Immunological evidence for the presence of NADPH Oxidase components in lens tissue
Lens homogenates (both soluble and insoluble) obtained from rat, monkey, and human lenses were probed for the presence of components of NADPH oxidase by western blot analysis using antibodies raised against recombinant subunits of human NADPH oxidase. A and B depict immunoreactivity against the p67phox and p47phox antibodies, respectively. In A and B, lanes 1 to 8 represent mouse soluble, rat soluble, monkey soluble, human soluble, rat insoluble, monkey insoluble, human insoluble lens fractions, and human neutrophil lysate, respectively. Human neutrophil lysate was used as a positive control for the expression of NADPH oxidase components. While mouse lens insoluble fraction showed positive immunoreactivity to the p67phox antibody, p47phox antibody did not reveal a clear immunoreactivity (data not shown).